
ヒドロキシプロピル Betadex HPBCD は薬物の溶解性とバイオアベイラビリティを強化します
CAS 番号: 128446-35-5
製品カテゴリー: 医薬品賦形剤
外観:白色またはほぼ白色の非晶質粉末
水溶性:水によく溶けます。
用途:医薬品製剤、注射剤、経口剤、外用剤
品質基準: USP / EP / ChP 医薬品グレード
Hydroxypropyl Betadex (HPβCD) is a high-purity pharmaceutical excipient widely used to improve the solubility, stability, and bioavailability of active pharmaceutical ingredients (APIs). It is commonly applied in drug formulation, cosmetics, and veterinary products.
As a modified cyclodextrin, HPβCD forms inclusion complexes with poorly soluble compounds, enabling better dissolution and enhanced formulation performance without altering the chemical structure of the API.
Hydroxypropyl Betadex HPBCD complies fully with United States Pharmacopeia (USP) and European Pharmacopoeia (EP) standards. Hydroxypropyl Betadex is suitable for various dosage forms including injectables, oral liquids, and topical applications. Hydroxypropyl Betadex manufactured under strict GMP conditions guarantees batch-to-batch consistency and predictable formulation performance.
Hydroxypropyl Betadex is widely chosen by formulators for research, scale-up, and product innovation projects. Hydroxypropyl Betadex provides a trusted solution for research and development teams seeking safe and effective excipients.
✔ Enhances drug solubility and bioavailability
✔ Improves stability of sensitive APIs
✔ Suitable for injectables, oral, and topical formulations
✔ Excellent safety profile and low toxicity
✔ Compatible with a wide range of pharmaceutical ingredients

Hydroxypropyl Betadex (HPβCD) is widely used as a pharmaceutical excipient to enhance the solubility, stability, and bioavailability of poorly water-soluble active pharmaceutical ingredients (APIs). By forming inclusion complexes, HPβCD enables more effective drug delivery across multiple dosage forms and administration routes.
HPβCD is commonly used in parenteral formulations to solubilize poorly soluble drugs and improve formulation stability. It helps reduce precipitation and ensures safe and effective delivery of APIs in aqueous solutions.
In oral formulations, HPβCD enhances dissolution rates and improves bioavailability. It is frequently applied in tablets, capsules, and oral liquids to support consistent drug absorption.
HPβCD is used in eye drop formulations to increase drug solubility while maintaining low irritation and high safety. It contributes to improved drug availability in ocular tissues.
HPβCD supports drug delivery through nasal sprays and inhalation systems by enhancing solubility and promoting rapid absorption through mucosal membranes.
HPβCD is also widely used in veterinary medicine to improve formulation performance and drug absorption in animal health products.
With over 27 years of manufacturing experience, Xi’an Deli Biochemical specializes in cyclodextrin production, including β-Cyclodextrin, Hydroxypropyl Betadex, and SBECD. Our products are supplied globally for pharmaceutical, veterinary, and cosmetic applications.
Xi’an Deli Biochemical provides technical support and regulatory documentation, including COA, DMF, and TDS, to support global regulatory submissions. Hydroxypropyl Betadex from Xi’an Deli Biochemical helps customers develop formulations efficiently and expand into international markets with confidence in product quality and compliance.
Our Hydroxypropyl Betadex (HPBCD) meets the requirements of major pharmacopeias, including USP, EP, and ChP. It is produced under a well-controlled manufacturing process to ensure consistent quality and reliable performance. This product is suitable for formulation development, scale-up, and various drug delivery applications.
| Item | Specification |
|---|---|
| Appearance | White or almost white amorphous powder |
| Solubility | Freely soluble in water |
| pH (1% solution) | 5.0 – 7.5 |
| Degree of Substitution | 0.4 ~ 1.5 |
| Loss on Drying | ≤ 5.0% |
| Heavy Metals | Complies with USP/EP |
|
Packaging |
500g/bag; 1kg/bag; 10 kg/drum |
|
Storage |
Store in a cool and dry place. |
| Shelf Life |
36 months when properly stored |
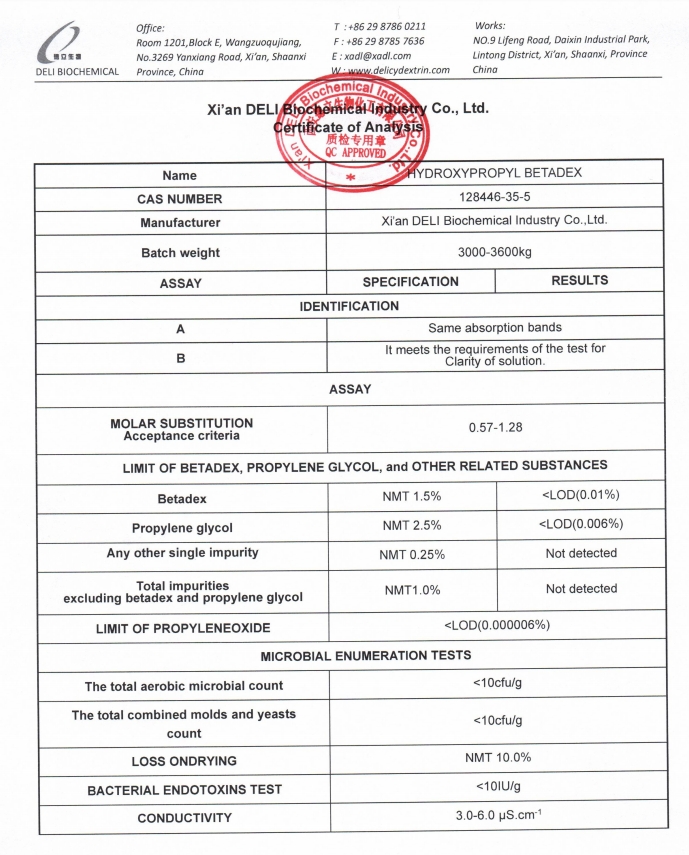
A detailed Certificate of Analysis (COA) is available upon request. Please refer to the COA image below for specification details.
Choosing Hydroxypropyl Betadex HPBCD Pharmacopeia Grade from Xi’an Deli Biochemical ensures collaboration with a reliable partner dedicated to quality, compliance, and innovation. Hydroxypropyl Betadex is available for free sample testing, allowing customers to evaluate Hydroxypropyl Betadex before scaling up production.
Hydroxypropyl Betadex has strong global recognition and extensive regulatory acceptance. Xi’an Deli Biochemical ensures Hydroxypropyl Betadex meets the highest standards required for pharmaceutical and cosmetic formulations. Hydroxypropyl Betadex helps customers achieve innovative, safe, and effective product development outcomes efficiently.
✔ High purity and consistent quality
✔ Full regulatory support (DMF available)
✔ Free samples for evaluation
✔ Reliable supply and technical assistance
Our Hydroxypropyl Betadex helps customers accelerate formulation development and successfully enter international markets.


1. What is Hydroxypropyl Betadex used for?
Hydroxypropyl Betadex is used as a pharmaceutical excipient to enhance the solubility, stability, and bioavailability of poorly soluble drugs.
2. Is HPBCD safe for injectable formulations?
Yes. Pharmaceutical-grade HPBCD is widely used in injectable drug formulations and is included in several approved pharmaceutical products.
3. How does HPBCD improve drug solubility?
HPBCD forms inclusion complexes with hydrophobic drug molecules, allowing them to dissolve more easily in aqueous environments.
4. What is the difference between β-cyclodextrin and HPBCD?
HPBCD is a modified form of β-cyclodextrin with hydroxypropyl groups that significantly improve water solubility and reduce toxicity.
5. What industries use Hydroxypropyl Betadex?
It is mainly used in pharmaceutical formulations, veterinary drugs, and advanced drug delivery systems.
Contact us today to request a free sample, COA, or technical documentation. Our team will support your formulation development and regulatory needs.
